Prostate Cancer: What Every Patient Should Know
Toby Soward, B.S.R.T. (R)
Elaine Halesey, Ed.D., R.T. (R)(QM)
Misericordia University
Medical Imaging Department
301 Lake Street
Dallas, PA 18612
(570) 674-6480 phone
(570) 67403052 fax
Contact Author: Elaine Halesey
This manuscript contains statistics relative to prostate cancer as well as a review of the related literature. It also provides information on the different types of treatment options for prostate cancer. Most importantly, it may serve as a reference for current or future patients undergoing treatment for prostate cancer as it describes the process a patient may experience.
TITLE SECTION
Material Title: Prostate Cancer: What Every Patient Should Know
Type of Material: Literature Review/Original Paper
Authors: Toby Soward, B.S.R.T. (R)
Elaine Halesey, Ed.D., R.T. (R)(QM)
Professional Affiliation Misericordia University
& Address: Medical Imaging Department
301 Lake Street
Dallas, PA 18612
Presentation at Scientific Meeting: Not Applicable
Abstract
Prostate cancer (PCA) is an uncontrolled malignant neoplasm of the prostate gland. It is “the second most commonly diagnosed malignancy in men,” and accounted for an estimated 30,200 deaths in 2002 (Gray, 2002, p. 151). Risk for PCA rises drastically with age, while white males are second only to African American males at risk. Exact causes are not known, however, family history, hormone levels, and diet are all thought to be contributing factors (Gray, 2002).
The American Cancer Society (ACS) estimated the number of men diagnosed in 2006 to be 234,460, and would result in 27,350 deaths (ACS, 2006, p. 4). Current statistics indicate that, “approximately 186,000 American men will be diagnosed with PCA and 28,600 deaths will occur (Up to Date, 2008). Once deemed necessary, Ultra Sound (US), Computed Tomography (CT), Positron Emission Tomography (PET), and Magnetic Resonance Imaging (MRI) and its counterparts such as Magnetic Resonance Stereoscopic Imaging (MRSI) have all been used to visualize its progress. Many treatments exist, including prostatectomy, radiotherapy (external and internal), cryosurgical ablation, hormone therapy, and chemotherapy. If the cancer is found in its early stages, survivability can be as high as 94%, while the five-year survivability in later stages is only 31% (Gray, 2002). Though data are important, it is vital to understand the patient’s experience throughout progression of the disease, as their experience is an important part of their journey to recovery.
Key Words:
Cancer
Intensity Modulated Radiation Therapy (IMRT)
Magnetic Resonance Spectroscopic Imaging (MRSI)
Multi-Leaf Collimators
Prostate
Radiation Therapy
Introduction
Much information exists regarding facts, figures, and overall progression of prostate cancer (PCA). Also important is what a patient being treated for PCA can expect. The purpose of this qualitative research is to share factual information regarding PCA, and more importantly, to provide patients who may undergo treatment for PCA with a better understanding of what they can expect. In addition, this article may serve as a resource for both educators and students of radiation therapy as it is important they, too, understand types of treatment and process to better serve their patients.
The prostate gland is one of the male’s accessory sex organs, roughly the size of a walnut. It is located anterior to the rectum, and inferior to the bladder, surrounding the urethra (National Kidney and Urologic Diseases Information Clearinghouse [NKUDIC], 2006). It produces fluids that both neutralize the acidity of the male urethra and female vagina, and nourish the male’s sperm once deposited into the female (NKUDIC, 2006). PCA is an uncontrolled malignant neoplasm of the prostate gland, arising from one of the prostate’s four zones. The four zones of the prostate are as follows: transitional zone, central zone, anterior fibromuscular stroma (all rarely sources of PCA), and the peripheral zone (origin of most PCA) (Gray, 2002). “Adenocarcinoma of the prostate accounts for more than 90% of all prostate cancers,” while mucinous adenocarcinomas, small cell, and transitional cell tumors are relatively rare (Gray, 2002, p. 153). This means that most prostate cancer arises from one place in the prostate and will continually grow, but will not function properly due to a defect of some kind. If these cells are not removed or killed, they will eventually spread throughout the body (metastasize), invading many of the body’s organs, rendering them useless.
Literature Review
Risk Factors and Demographics
PCA is “the second most commonly diagnosed malignancy in men,” with an estimated 189,000 men diagnosed in 2002 (Gray, 2002, p. 151). In a comparison of studies, “It [was] the second leading cause of cancer deaths in American males and accounted for an estimated 30,200 deaths in 2002 and surpassed only by lung cancer,” (Gray, 2002, p. 151). In contrast, the American Cancer Society (ACS) estimated the number of men diagnosed in 2006 to be 234,460, and would result in 27,350 deaths (ACS, 2006, p. 4). It should be noted that although more cases of PCA were estimated in 2006 than 2002, fewer deaths were estimated. The probability for a man to develop PCA in his lifetime is roughly .17, or a 1 in 6 chance (ACS, 2006, p. 13). Current statistics indicate that, “approximately 186,000 American men will be diagnosed with PCA and 28,600 deaths will occur (Up to Date, 2008).
Experts believe that there are many risks for developing PCA. Race is a large factor in the development of PCA, as African Americans are more likely to develop it compared to Anglo Americans, with a ratio of 1.6 to 1 (ACS, 2006). Using this statistic, 160,000 African Americans would be diagnosed for every 100,000 Anglo Americans diagnosed with PCA. The mortality rate for African Americans versus Anglo Americans is 2.4 to 1, or 240,000 African American versus 100,00 Anglo American deaths (ACS, 2006). Geriatrics is the largest group at risk for PCA. The incidence for PCA in various age groups is as follows: birth-39 (1 in 10,149), 40-59 (1 in 38), 60-69 (1 in 14) and 70-death (1 in 7) (ACS, 2006). These statistics indicate that a 60 year old is almost 725 times more likely to develop PCA than someone 39 years old; a 70 year old is double that, or almost 1450 times as likely.
There are also a few less influential factors affecting the population with PCA. Family history may contribute to 5-10% of PCA (ACS, 2006). Diets high in saturated fat and obesity are also factors (ACS, 2006). The fact that PCA is more common in North America and northwestern Europe (arguably more obese areas) than in Asia and South America corroborates the obesity theory. Based on this information, a 70-year-old African American man who is obese and whose father has PCA would almost certainly develop it as well.
There are no known definitive causes for PCA (Gray, 2002). The ACS surmises that diets high in saturated fat—especially red meat and butter (Gray, 2002)—could be a cause (2006). Gray also indicates that environmental factors play a part, as the incidence of PCA in Japanese men increases when they move to America from Japan (2002). High levels of serum testosterone and insulin-like growth factor-1 are also thought to increase risk, although the exact cause is unclear (Gray, 2002). Gray states that vasectomy is not a viable cause of PCA (2002). Despite the many possible causes of PCA, there are several contraindications. Diets high in nuts, some types of meat, vegetables, and eggs may reduce PCA risk due to their content of selenium (Gray, 2002). Additionally, lycopene, found in tomatoes, grapefruit, and watermelon, may reduce the risk of PCA.
Detecting PCA
Before any imaging procedures of the prostate are completed, a physician usually performs and/or orders two other tests. First, a digital rectal exam is performed to evaluate the size of the prostate (Jani & Hellman, 2003). If the prostate is enlarged, a Prostate Specific Antigen (PSA) test is usually performed. Gray states that, “PSA is a protein normally produced by the prostate gland” and, “mild to moderate elevation of the PSA may indicate PCA, [while] a marked elevation is highly suspicious of PCA,” (2002, p. 157). If PSA levels are elevated, additional imaging may be performed.
Michael Burke, M.D., is a radiation oncologist and Medical Director of Radiation Oncology practicing in the Scranton, PA area. Certified by the American Board of Radiology, he has served as Medical Director at several oncology centers including his current position. Burke recommends that every male over 50 have both a yearly digital rectal exam and regular PSA tests, despite any question of the cost-effectiveness of the test (personal communication, November 3, 2008). In cases where the men are African American or genetically predisposed to PCA, testing is recommended at age 40. In some cases, if the digital rectal exam or PSA test is inconclusive, imaging of the prostate will be done.
Prostate Imaging
One imaging modality used for the prostate is trans-rectal ultra sound (TRUS). Rifkin, Dahnert, and Kurtz state “by placing a transducer in the rectum close to the prostate, it is possible to use high-resolution transducers with sharply focused near fields” (1990, p. 691). Sonography alone may not be good enough, however, as Halpern and Strup state, “Even among patients with an elevated PSA level (≥10 ng/ml) who might be expected to have larger tumor burdens, sonography showed low sensitivity. Furthermore, sonography failed to detect 35 of 99 cancerous foci with a Gleason score of 7 or higher” (2000, p. 626).
Positron Emission Tomography (PET) is the modality of choice to visualize PCA, as evidenced by the statement, “functional imaging with positron emission tomography (PET) will detect malignant tumors in the prostate and/or prostate bed, as well as possibly help determine tumor aggressiveness” (Huber, Moses, Pouliot, & Hsu, 2005, p. 1). One of the emerging trends in PET is to use the radiopharmaceutical choline to visualize the prostate. In a study evaluating the use of choline, it was found that when using this chemical, “the urinary activity was negligible and tumor uptake was marked” (Hara, Kosaka, & Kishi, 1998, p. 990). Additionally, the study concluded “prostate cancer and its local metastasis were visualized clearly in PET using choline” (Hara et al., 1998, p. 990).
Three-Dimensional Computed Tomography (3D CT) is also used to visualize the prostate. Another approach that is becoming more common is using multiple modalities at the same time to visualize the prostate. Three-dimensional CT and ultrasound (US) are used in the treatment planning for brachytherapy (CET, n. d.). Another combination is PET and US; a type of fusion imaging. US is being used in conjunction with PET to provide better detail to anatomy; the uptake is so great that other anatomy is obscured with PET alone (Huber et al., 2005). They also state:
Ultrasound imaging with a trans-rectal probe provides anatomical detail in the prostate region that can be co-registered with the sensitive functional information from the PET imaging. Imaging the prostate with both PET and trans-rectal ultrasound (TRUS) will help determine the location of any cancer within the prostate region. This dual-modality imaging should help provide better detection and treatment of prostate cancer (Huber et al., 2005, p. 1).
Magnetic Resonance Spectroscopic Imaging (MRSI) is a modality that is becoming instrumental in the imaging of the prostate. MRSI combines the superior soft tissue imaging capability of MRI with spectroscopy. Spectroscopy, with regard to MRI, is “a special technique used for characterization of the biochemistry of tumors, infarcts, and other pathology” (Radiology Consultants of North Dallas [RCND], n.d.). The basic idea of MRSI is to not actually image the organ itself, but the chemicals within it. Prostate cancer is known to have more of a certain chemical and less of another; being able to see those chemicals allows physicians to determine more accurately the presence of cancer. MRI alone has demonstrated an admirable 78% sensitivity (ability to diagnose the disease) to PCA, but only 55% specificity (not having a false positive) (Swanson, Vigneron, Tran & Kurhanewicz, 2001). With the addition of spectroscopic imaging, however, both numbers dramatically rise. In a study of 53 biopsy-proven PCA patients, MRSI resulted in 95% sensitivity and 91% specificity (Swanson et al., 2001). For comparison, Prostate Specific Antigen (PSA) testing is also used to diagnose PCA; having a sensitivity of 80% and a specificity of 83 % (Lujan, Paez, Llanes, Miravalles & Berenguer, 2001). Sonography is another modality for diagnosis, having a sensitivity of 44% and a specificity of 74% (Halpern & Strup, 2000). Most surprisingly, Positron Emission Tomography (PET), specifically testing choline levels, has a sensitivity of 91% and a specificity of 50% (Rinnab et al., 2007). When considering the sensitivity and specificity of the three prominent diagnostic tools for PCA, it is clear that MRSI is the superior modality. The only shortcomings of MRSI are that of traditional MRI; claustrophobia and the use of the endorectal coil.
Staging and Grading of PCA
The following chart describes the staging and grading of PCA:
Staging (The Prostate Program, n.d.)
T1c (PSA
detected)
T2a (small nodule)
T2b (larger nodule)
T3 (very large nodule with probable extension outside of
the prostate)
T4 (prostate cancer growing into adjacent organ such as
the bladder)
Grading (Gleason Score) (The Prostate Program, n.d.)
2 – 4 slow growing (well differentiated)
5 – 7 intermediate (moderately differentiated)
8 – 10 fast growing (poorly differentiated)
Treatment
Watchful waiting.
Watchful waiting indicates that no treatment will be given to the patient unless they begin to show symptoms of metastatic disease (Pirtskhalaishvili, Hrebinko, & Nelson, 2001). The patient’s PSA levels are continually monitored, and treatment options are revaluated based on the patient’s condition and the progression of the disease (Pirtskhalaishvili et al., 2001). Watchful waiting is generally used when the PCA is detected incidentally, and the patient’s life expectancy is less than ten years (Pirtskhalaishvili et al., 2001). This is an accepted treatment method because the cancer is not expected to become a problem before the patient would die of other causes (Pirtskhalaishvili et al., 2001).
Radical prostatectomy.
Radical prostatectomy is the “removal of the prostate, seminal vesicles, and adjacent tissues including overlying fat and pelvic sidewall (Gray, 2002). There are different types of prostatectomy, based on approach. The retropubic prostatectomy uses an incision from the umbilicus to the symphysis pubis, and is the preferred method because it is usually able to spare the nerves needed for sexual function (Gray, 2002). A non-nerve sparing procedure is the perineal prostatectomy, usually done when the tumor invades local tissues (Gray, 2002). To complete the perineal prostatectomy, a curvilinear incision is placed in the perineum from one ishial tuberosity to the other.
External and internal radiotherapy.
External radiotherapy is what most people think of as radiation therapy. It uses high-energy radiation in excess of 70 Gray (Gy) (a unit of measure of radiation) to kill the cancer cells. This level of radiation is very toxic to surrounding tissues, therefore the therapy is given in the course of many weeks, and care is taken to only deliver radiation to the tumor (Gray, 2002).
Internal radiation therapy is known as brachytherapy. In brachytherapy, radioactive seeds encased in titanium are surgically implanted into the prostate (Gray, 2002). The seeds may either be placed into the patient temporarily or permanently (Gray, 2002). Both types of radiotherapy have side affects to include rectal bleeding, urinary problems, and sexual problems (Pirtskhalaishvili et al., 2001).
Cryosurgical ablation.
This method uses trans-rectal ultrasound to guide catheters into the prostate, through which liquid nitrogen will be introduced (Pirtskhalaishvili et al., 2001). This method is problematic, however, when attempting to treat the superior prostate or seminal vesicles, as the amount of freezing required tends to damage surrounding tissues (Pirtskhalaishvili et al., 2001). Impotence is generally more common in cryosurgical ablation than in prostatectomy, however, long-term results of efficacy and side effects are not yet available (Pirtskhalaishvili et al., 2001).
Hormone therapy.
Testosterone levels and prostate cancer are related. One study found that “androgens are implicated in the growth regulation of the healthy gland, as well as the pathenogenesis of prostate carcinoma” (Pirtskhalaishvili et al., 2001, p. 299). One method to reduce androgens is bilateral orchiectomy (Pirtskhalaishvili et al., 2001). Luteinizing hormone and adrenal androgen are also hormones that are lowered, and anti-androgens are administered via medications (Pirtskhalaishvili et al., 2001). Although very effective at retarding the growth of PCA, hormone therapy is palliative, not curative (Pirtskhalaishvili et al., 2001).
Chemotherapy.
Once hormone therapy is no longer effective, chemotherapy is used to treat PCA. It uses the drugs estramustine, mitoxantrone, taxanes, and suramin in an attempt to further reduce hormone levels, increased white blood cell activity, and decreased tumor cell activity (Pirtskhalaishvili et al., 2001).
Burke states he prefers to use a form of external radiation therapy known as Intensity Modulated Radiation Therapy (IMRT) in most cases of PCA (personal communication, November 3, 2008). This form of therapy allows the radiation beam to be shaped specifically to each individual patient’s anatomy. IMRT, while much more effective at destroying cancer cells than conventional radiation therapy, is also more expensive. However, IMRT is not as effective at reducing tumors as another form of therapy that uses IMRT compensators. These compensators are a series of metal blocks that shape the radiation beam precisely to the patient’s specific anatomy and tumor. This type of therapy that involves the metal blocks is the most expensive of the three: conventional radiation therapy; IMRT; or IMRT with compensators. Therefore many patients and physicians choose to use IMRT for the treatment of PCA, as it is the best balance between cost and effectiveness in treating PCA (Burke, personal communication, November 3, 2008).
Discussion
The Patient’s Experience
While it is important to understand the facts of PCA, it is imperative that the experiences of the patients are not overlooked. It is the focus of the remainder of this article to describe the experience of a patient who is being treated for PCA.
When a patient first tests positive for PCA, they will meet with a radiation oncologist to discuss their options. The oncologist will discuss the patient’s stage and grade of tumor, as well as their options for treatment. If the stage and grade of the tumor is high, and the patient is over 70, no treatment may be recommended; whereas if the same tumor were present in a 30-year-old patient, radical prostatectomy as well as IMRT may be used (Burke, personal communication, November 3, 2008). Some physicians prefer IMRT in many cases of PCA because it is very effective at destroying cancer, can be used in conjunction with prostatectomy, and has comparable side effects to other forms of therapy. Upon consulting with the physician, the patient is asked to read and sign a consent form for the pertinent body part that will be treated. The oncologist, patient, and a witness must sign the consent form (Appendix A).
Simulation Procedure
Before treatment can begin, the patient must go through a simulation, or sim, for short. The purpose of this sim is to provide an accurate location of the tumor and the patient’s anatomy so a treatment plan can be tailored specifically to them. The sim also ensures that the patient is placed in the same exact position every time they are treated, as this is vital to the efficacy of the IMRT treatment. Once the sim is completed, the oncologist, a dosimetrist (a person who designs treatment plans), and a medical physicist collaborate on a therapy plan that will be administered to the patient. To ensure the utmost efficacy of the treatment, the sim, treatment, and all pertinent calculations are performed with the utmost attention to detail.
There is specific preparation the patient must undergo to ensure their internal anatomy is in the same location every time they are treated, as the bladder and rectum move constantly within the pelvic cavity. This prep includes taking laxatives to empty the rectum, then voiding the bladder, followed by imbibing a specific amount of liquid in an attempt to fill the bladder to a uniform size every time the patient is treated (Appendix B). The patient undresses from the waist down, donning a hospital gown. While the patient is waiting for the sim to begin, radiation therapists (people who position patients and administer the treatment plan) prepare the sim room by calibrating the CT lasers to a set of external lasers that will be used during the tattooing of the patient. It is imperative that these lasers are calibrated within a tolerance of 2mm so the tumor can be targeted as accurately as possible.
When the patient enters the sim room, they are told to lie on the CT table. A pillowcase or sheet is placed over their groin, and the gown is raised above the waist. This is to ensure the gown is not bunched behind the patient’s legs, making it more difficult to reproduce the patient’s exact position during treatment. Next, a vacloc bag (Appendix C1) is placed under their legs, and the patient is given a plastic ring to hold on their chest to keep their hands and arms out of the treatment area.
All appendices may be found at the end of this document, immediately following the list of references also at the end. The vacloc bag contains styrofoam pellets and air. When the air is vacuumed out of the bag, it conforms perfectly to the patient’s legs. This specific vacloc bag will be used for the patient for the duration of their treatment to ensure they are positioned on the table in the same manner every time. After the vacloc process is formed, the patient is placed partially into the CT machine and the external lasers are placed in what the radiation therapist determines a proper site for administering the tattoos (Appendix C 2&3). These lasers intersect in three different locations on the patient: the pubic area, and the sides of both upper thighs. At this point, the physician checks the patient’s position and a CT scan is performed, placing metal markers (BB’s) at the intersections of the lasers so they can be visualized on the CT scan (Appendix D 1&2). If the physician is satisfied with the scan, the patient is then tattooed (Appendix D 3-6).
Because the purpose of the tattoos is to ensure proper placement of the patient every time they are treated, they correspond exactly to where the BB’s were placed for the CT scan. It will be these tattoos that the therapist aligns with the lasers in the treatment room upon administering the radiation. It cannot be overstressed that proper placement of these tattoos, according to the markers on the scan, is vital. The idea of the tattoo is generally worse for the patient than the reality of getting it. Many patients are afraid of needles, especially since the needle for the tattoo is a large (18) gauge. However, this tattoo is not the same as one may commonly think; it is simply a prick by a needle with black ink on the end leaving a very small permanent dot on the patient. After the tattoos are placed, photos are taken of the entire setup, as well as close ups of the tattoos. Many patients feel very self-conscious during this final portion of the sim because they feel vulnerable and exposed having photos taken of them. Elderly patients have an especially difficult time lying on the hard table during this procedure. In addition, they have had a full bladder for the entire 30-60 minute sim, and are uncomfortable, as dysuria and urinary and fecal incontinence are very common in patients with PCA. Once the sim is complete, the patient is sent home while their specialized treatment is created by the aforementioned professionals.
Treatment Planning
There are many facets of the treatment planning process that do not fall under the scope of this document. What is important to understand about the treatment planning is the determination of the isocenter of the treatment area, and its relationship to the tattoos that were placed on the patient during the sim. The isocenter of the treatment area is simply the very center of the area that the physician prescribes to be treated with radiation. This is not necessarily just the prostate, as surrounding areas are usually treated prophylactically. This area still has to be small, however, as unwanted radiation exposure to the bladder and rectum is avoided much as possible. After the isocenter of the treatment area is defined by the oncologist, the dosimetrist provides the location of the isocenter relative to the tattoo marks on the patient. Recall that the tattoos were placed by the radiation therapists prior to knowing exactly where the isocenter would eventually be, so it is necessary to know how far to shift the patient from the tattoos to the isocenter of the treatment area.
Treatment Delivery
Once the treatment plan is completed, which can take days to weeks, the patient is contacted to return. At this point, the patient is usually apprehensive about their treatment, as they have had a good deal of time to think about it. The prep for the patient includes first emptying the bladder at a predetermined time before their treatment. They are then required to drink a specified amount of water prior to the treatment in an attempt to have the same amount of liquid in the bladder each time of treatment. It is imperative the bladder is full for the treatment because it greatly reduces the chance of toxicity to the bladder. Additionally, it places the bladder in roughly the same position every day. The patient is usually very uncomfortable at this time, because they have a full bladder, and are usually incontinent. The patient then removes all clothes from the waist down, donning a hospital gown.
Next, the patient is called into the treatment room and greeted by two to three therapists. The patient is placed on the treatment table in the exact manner as they were for the sim, including the vacloc bag, and holding the ring on their chest. It is imperative the patient remain perfectly still from this point forward. This is very difficult for many patients, as they many have urinary and fecal incontinence, as previously noted. Once on the treatment table, a sheet is again placed over the groin, the gown opened, and the tattoos exposed. The therapist aligns these tattoos to the lasers in the room, which represent the isocenter of the treatment beam of the machine in the room (Appendix E 1&2). At this point, the therapists shift the patient from the tattoo marks to the isocenter of the treatment area that has been provided by the oncologist and dosimetrist (Appendix E 3&4). Once the shifts are completed, BB’s are placed on the new intersections of the lasers, and the therapist spins the table that the patient is on to place them in a CT scanner, also in the room. This is very similar to the sim that the patient already experienced. The purpose of this scan is so the oncologist can determine the exact location of the prostate at that specific time. From this scan, the oncologist may choose to shift the position of the patient so the isocenter of the treatment area is in the exact location as the isocenter of the treatment beam. Once the patient is shifted again, the treatment begins. By this time, the patient has been on the treatment table for approximately 10 to 15 minutes.
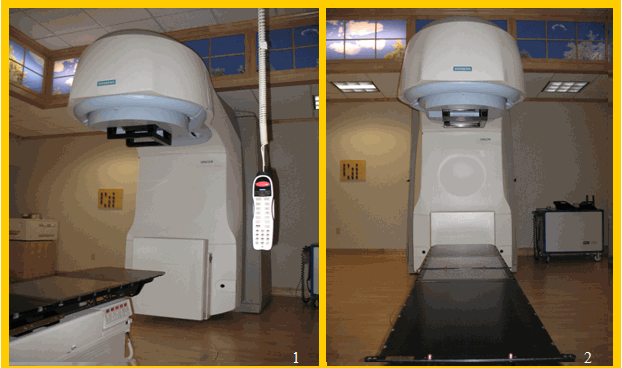
The entire treatment could take up to 25 minutes, as there are usually five different angles from which the radiation beam enters the patient. IMRT treatments use small lead blockers known as Multi-Leaf Collimators (MLC’s) in the head of the machine that shape the radiation beam, and these leaves must also move during the treatment (Appendix F 1-4). During this treatment, the patient feels nothing.
When the treatment is over, the patient is assisted from their position. At this point, the patient usually experiences a great deal of embarrassment, as they may have not been able to control their urinary or defecating functions. The patient is then escorted to the changing room where they can clean up, change and go home. A typical patient with PCA will have about 50 of these treatments. The complications they may experience from the treatments are dysuria, increased urinary and fecal incontinence, skin irritation, and possibly impotence. The patient will follow up with the oncologist a few weeks after their final treatment to discuss any complications they may be having from the treatment. Then, the patient usually comes in for follow up appointments every six months to have a digital rectal exam, providing they have not had a prostatectomy. Periodic PSA levels are obtained as well.
Overall Success of Treatment
A study by the Cleveland Clinic suggested the success rates of other IMRT treatments. They found that men with PCA who were treated with IMRT had a six-year survival rate of 100% if they were low risk, meaning their tumor stage was T1-T2, and their Gleason grade was ≤6 (Mundt & Roeske, 2005). In high-risk patients with stage T3 tumors with grades ≥6, the 6-year survival was still 81% (Mundt & Roeske, 2005). All of this was accomplished without the patients experiencing low amounts of acute toxicity to adjacent organs (Mundt & Roeske, 2005). These figures were with patients that had not undergone radical prostatectomies; meaning their survival rates may have been higher in the high-risk group had they chosen to have prostatectomies.
Conclusion
As demonstrated, there are many factors concerning PCA that one must understand to fully comprehend the disease. The demographics affected, diagnostic studies used, treatments, and morbidity and mortality rates; all play an important role in fully understanding PCA. However, many articles are remiss by not including very important information; the patient’s experience. It is our hope this document not only educates the reader about the facts and treatment options for PCA, but provides current and/or future patients with a better understanding of what they can expect on their personal journey.
References
American Cancer Society (ACS). (2006). Cancer facts & figures 2006. Retrieved November 27, 2007 from http://www.cancer.org/downloads/STT/CAFF2006PWSecured.pdf
California Endocurietherapy Cancer Center (CET). (n. d.). What is brachytherapy? Retrieved September 21, 2007 from http://www.cetmc.com/prostate.html#ct_based_dosimetry
Gray, M. (2002). A prostate cancer primer. Urologic Nursing, 22(3), 151.
Halpern, E. J., & Strup, S. E. (2000). Using gray scale, color and power doppler sonography to detect prostatic cancer. American Journal of Roentgenology, 174, 623–627.
Hara, T., Kosaka, N., & Kishi, H. (1998). PET imaging of prostate cancer using
carbon-11-choline. Journal of Nuclear Medicine, 39, 990-995.
Huber, J. S., Moses, W. W., Pouliot, J., & Hsu, I. C. (2005). Dual-modality PET/ultrasound imaging of the prostate. Retrieved November 27, 2007 from http://www.osti.gov/bridge/servlets/purl/862086-WIShew/862086.PDF
Jani, A. B., & Hellman, S. (2003). Early prostate cancer: Clinical decision-making. Lancet, 361(9362), 1045.
Lujan, M., Paez, A., Llanes, L., Miravalles, E., & Berenguer, A. (2001). Prostate specific antigen density. Is there a role for this parameter when screening for prostate cancer? Prostate Cancer & Prostatic Diseases, 4(3), 146.
Mundt, A. J., & Roeske, J. C. (2005). Intensity modulated radiation therapy: A clinical
perspective. Hamilton, Ontario: BC Decker Inc.
National Kidney and Urologic Diseases Information Clearinghouse (NKUDIC). (2006). The prostate gland. Retrieved November 27, 2007 from http://kidney.niddk.nih.gov/kudiseases/pubs/prostateenlargement/#gland
Pirtskhalaishvili, G., Hrebinko, R. L. & Nelson, J. B. (2001). The treatment of prostate cancer: An overview of current options. Cancer Practice, 9(6), 295-306.
Prostateline. (2009). Prostate gland anatomy and physiology. Retrieved February 19, 2009 from:
http://www.prostateline.com/prostate-cancer/anatomy-andphysiology?itemId=2617452&nav=yes
Radiology Consultants of North Dallas (RCND). (n.d.). Latest developments: MRI spectroscopy
(MRS). Retrieved April 21, 2008 from http://www.rcnd.com/MRS.html
Rifkin, M. D., Dahnert, W., & Kurtz, A. B. (1990). State of the art: Endorectal sonography of the prostate gland. American Journal of Roentgenology, 154, 691–700.
Rinnab, L., Mottaghy, F. M., Blumstein, N. M., Reske, S. N., Hautmann, R. E., Hohl, K., et al. (2007). Evaluation of [11C]-choline positron-emission/computed tomography in patients with increasing prostate-specific antigen levels after primary treatment for prostate cancer. BJU International, 100(4), 786-793.
Swanson, M., Vigneron, D., Tran, T., & Kurhanewicz, J. (2001). Magnetic resonance imaging and spectroscopic imaging of prostate cancer. Cancer Investigation, 19(5), 510.
The Prostate Program. (n.d.) Grading and staging. Retrieved August 7, 2008 from http://www.prostateprogram.org/prognostic.html
Up to Date. (2008). Overview of treatment for early prostate cancer. Retrieved November 19, 2008 from http://www.utdol.com/online/content/topic.do
Varian Medical Systems (VMS). (2008). Multi-leaf collimators. Retrieved November 25, 2008 from http://varian.mediaroom.com/index.php?s=13&cat=22&mode=gallery
Appendices
Appendix A …………………………………………………. Patient Consent Form
Appendix B………………………………………………….. Sim Prep Instructions
Appendix C…………………………………………………...VacLoc and Laser Placement
Appendix D…………………………………………………..BB’s and Tatooing
Appendix E……………………………………………………Isocenter of Beam & Shifts from Tattoos to Isocenter of Treatment Area
Appendix F……………………………………………………Linear Accelerator Treatment Machine and Multi-Leaf Collimators
![]()

(Reprinted with permission from Professional Radiation Oncology Partners, Scranton, PA)

Appendix C
(Images courtesy of Professional Radiation Oncology Partners, Scranton, PA)
Vacloc & Laser Placement
C.1.
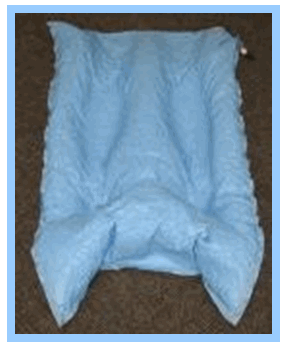
C. 2. & 3.

Appendix D
(Images courtesy of Professional Radiation Oncology Partners, Scranton, PA)
BB’s & Tattooing
D 1&2

D 3-6

Appendix E
(Images courtesy of Professional Radiation Oncology Partners, Scranton, PA)
Isocenter of Beam & Shifts from Tattoos to Isocenter of Treatment Area
 E
1&2
E
1&2
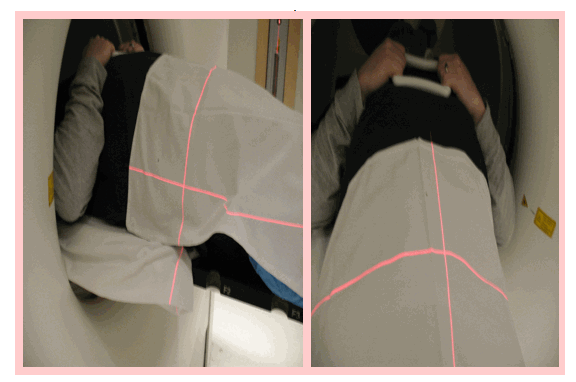 E
3&4
E
3&4
Appendix F
(Images 1-3 courtesy of Professional Radiation Oncology Partners, Scranton, PA)
Linear Accelerator (Treatment Machine) (1&2) & MLC’s (3&4)